-
Best Australian Pokies Best Payout
Play Free 777 Pokies Online
What Are the Best Australian Pokies to Win Big On
What Are the Best Online Pokies with Low Minimum Deposits for Real Money in Australia
New Australian Pokies Accepting PayID
What Are the Best Strategies to Win in Online Pokies with Bonus Rounds in Australia
Tips for Winning Top Australian Pokies
What Online Casinos Offer Free Play Pokies with Welcome Bonuses in Australia
Australian Slot Games
Payout Ratio Pokies with Welcome Bonus
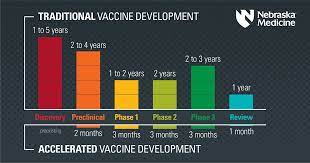
We cover in Week 10 the process pharmaceutical companies follow to develop and approve a new product. The COVID vaccines were approved (and researched) differently.
Week 10 the process pharmaceutical companies follow to develop
We cover in Week 10 the process pharmaceutical companies follow to develop and approve a new product. The COVID vaccines were approved (and researched) differently. This week, I expect you to research how the process was different for COVID vaccines and take a position as to what should change when approving new pharmaceutical products and what shouldn’t. Which exceptions were warranted for COVID vaccines, in your opinion, but should not become changes to the process for other products in your opinion?
To understand how the process was different, here is a Youtube video from Pfizer on how they developed the vaccine: https://www.youtube.com/watch?v=jbZUZ9JYNBE
And a video of a virtual talk a scientist involved in the development of the Oxford-AstraZeneca vaccine gave: http://www.bristol.ac.uk/blackwell/news/2021/apl-2021.html (the talk itself is ~30 minutes followed by a question period. There are questions about vaccine development around minute 39).
There are several COVID-19 vaccines validated for use by WHO (given Emergency Use Listing). The first mass vaccination programme started in early December 2020 and the number of vaccination doses administered is updated on a daily basis on the COVID-19 dashboard.
The WHO Emergency Use Listing process determines whether a product can be recommended for use based on all the available data on safety and efficacy and on its suitability in low- and middle-income countries. Vaccines are assessed to ensure they meet acceptable standards of quality, safety and efficacy using clinical trial data, manufacturing and quality control processes. The assessment weighs the threat posed by the emergency as well as the benefit that would accrue from the use of the product against any potential risks.
In line with their national regulations and legislation, countries have the autonomy to issue emergency use authorizations for any health product. Domestic emergency use authorizations are issued at the discretion of countries and not subject to WHO approval.
As of 12 January 2022, the following vaccines have obtained EUL:
- The Pfizer/BioNTech Comirnaty vaccine, 31 December 2020.
- The SII/COVISHIELD and AstraZeneca/AZD1222 vaccines, 16 February 2021.
- The Janssen/Ad26.COV 2.S vaccine developed by Johnson & Johnson, 12 March 2021.
- The Moderna COVID-19 vaccine (mRNA 1273), 30 April 2021.
- The Sinopharm COVID-19 vaccine, 7 May 2021.
- The Sinovac-CoronaVac vaccine, 1 June 2021.
- The Bharat Biotech BBV152 COVAXIN vaccine, 3 November 2021.
- The Covovax (NVX-CoV2373) vaccine, 17 December 2021.
- The Nuvaxovid (NVX-CoV2373) vaccine, 20 December 2021
WHO recommends that while vaccine supply is limited, the people at highest risk of COVID-19 are vaccinated first. This includes people who are more likely to get severe disease if they are infected (older persons and people with existing health conditions) and people who are more likely to be exposed to the virus (such as health workers). People who are pregnant have a higher risk of serious illness and preterm birth if they are infected with COVID-19, so WHO recommends that they are also prioritized for vaccination, once the first priority groups have been vaccinated.
If you live in a country where vaccines are available to more people beyond these priority groups, get vaccinated as soon as it is your turn.
WHO-authorized COVID-19 vaccines are safe for most people of 18 years and older, including those with pre-existing conditions of any kind such as auto-immune disorders. These conditions include hypertension, diabetes, asthma, pulmonary, liver and kidney disease, as well as chronic infections that are stable and controlled.
The Pfizer vaccine can be safely administered to children from 5 years of age. Both Moderna and Pfizer vaccines are licensed for use in children from 12 years of age.
Vaccine trials for the use of other COVID-19 vaccines in children and adolescents are ongoing and WHO will update its recommendations when the evidence or epidemiological situation justifies a change in policy.
Children and adolescents above 5 years of age with comorbidities that put them at significantly higher risk of serious COVID-19 may be offered vaccination alongside other high priority groups.
However, WHO recommends that countries should vaccinate healthy children only when high vaccine coverage with two doses has been achieved in higher priority-use groups, as identified in the WHO Prioritization Roadmap.
Attachments
