-
Best Australian Pokies Best Payout
Play Free 777 Pokies Online
What Are the Best Australian Pokies to Win Big On
What Are the Best Online Pokies with Low Minimum Deposits for Real Money in Australia
New Australian Pokies Accepting PayID
What Are the Best Strategies to Win in Online Pokies with Bonus Rounds in Australia
Tips for Winning Top Australian Pokies
What Online Casinos Offer Free Play Pokies with Welcome Bonuses in Australia
Australian Slot Games
Payout Ratio Pokies with Welcome Bonus
Moving chemical structures and NMR data from a preexisting word document into the University template. Numbering of Chemical structures based on the university conventions. These numbers appear under each compound — a chemist will understand this.
Moving chemical structures and NMR data
Below are a list of required tasks for my dissertation document. In general the document is a mix of synthetic chemistry and biology. Chapters 1-2 are based
on 3 peer reviewed American Chemical society publications. Chapter 3 Contains unpublished data.
The tasks required will include:
1) Moving chemical structures and NMR data from a preexisting word document into the University template.
2) Numbering of Chemical structures based on the university conventions. These numbers appear under each compound — a chemist will understand this. The university policy is that the 3rd compound in chapter 1 willappear as 1.3, the 8th compound in chapter 3 will be 3.8 etc.
3) General formatting of the document based on the university standard. This will include citations, data appendix etc.
Moving Chemical Structures and NMR Data from a Preexisting Word Document into the University Template
Do you have a chemistry lab report or research paper that you are working on? Are you struggling to format the chemical structures and NMR data correctly? Don’t worry, we can help! In this blog post, we will show you how to move your data from a preexisting word document into the University template. This process is relatively simple, and it will save you a lot of time and hassle in the long run. Let’s get started!
The first thing you need to do is open up your word document and locate the chemical structures and NMR data. Once you have found them, select all of the data and copy it. Next, open up the University template and create a new section for your chemical structures and NMR data. To do this, simply click on the “Insert” tab and then select “New Section.”
Once you have created a new section, paste your copied data into it. Now, you need to format the data so that it looks correct. To do this, go to the “Format” tab and select “Columns.” In the Columns menu, select “One Column.” This will ensure that all of your data is correctly formatted.
Now that your data is correctly formatted, you need to add the proper citations. To do this, go to the “References” tab and select “Add Citation.” In the Add Citation menu, select “Book” as the citation type.
Then, fill out all of the necessary information about the book. Once you have finished adding the citation, your data should be correctly formatted and ready to go!
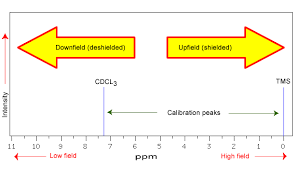
Nuclear precession, spin states, and the resonance condition
When a sample of an organic compound is sitting in a flask on a laboratory benchtop, the magnetic moments of its hydrogen atoms are randomly oriented. When the same sample is placed within the field of a very strong magnet in an NMR instrument (this field is referred to by NMR spectroscopists as the applied field, abbreviated B0 ) each hydrogen will assume one of two possible spin states.
In what is referred to as the +½ spin state, the hydrogen’s magnetic moment is aligned with the direction of B0, while in the -½ spin state it is aligned opposed to the direction of B0.
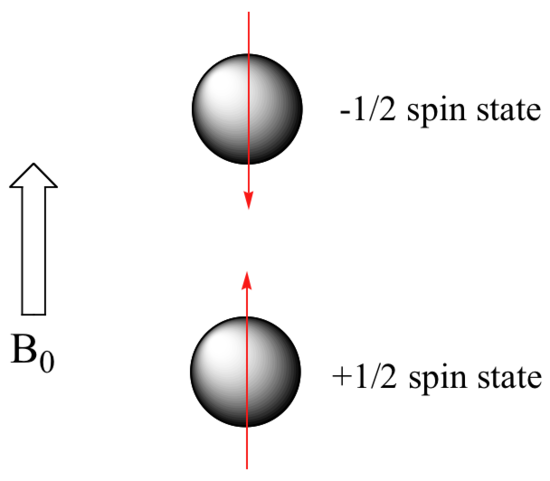
Because the +½ spin state is slightly lower in energy, in a large population of organic molecules slightly more than half of the hydrogen atoms will occupy this state, while slightly less than half will occupy the –½ state. The difference in energy between the two spin states increases with increasing strength of B0.This last statement is in italics because it is one of the key ideas in NMR spectroscopy, as we shall soon see.
At this point, we need to look a little more closely at how a proton spins in an applied magnetic field. You may recall playing with spinning tops as a child. When a top slows down a little and the spin axis is no longer completely vertical, it begins to exhibit precessional motion, as the spin axis rotates slowly around the vertical. In the same way, hydrogen atoms spinning in an applied magnetic field also exhibit precessional motion about a vertical axis.
It is this axis (which is either parallel or antiparallel to B0) that defines the proton’s magnetic moment. In the figure below, the proton is in the +1/2 spin state.
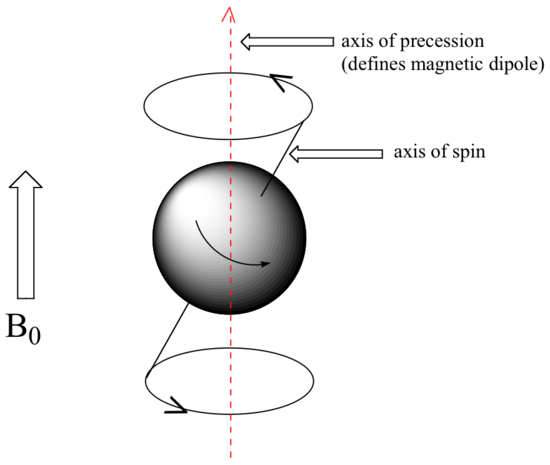
The frequency of precession (also called the Larmour frequency, abbreviated ωL) is simply the number of times per second that the proton precesses in a complete circle. A proton`s precessional frequency increases with the strength of B0.
If a proton that is precessing in an applied magnetic field is exposed to electromagnetic radiation of a frequency ν that matches its precessional frequency ωL, we have a condition called resonance. In the resonance condition, a proton in the lower-energy +½ spin state (aligned with B0) will transition (flip) to the higher energy –½ spin state (opposed to B0). In doing so, it will absorb radiation at this resonance frequency ν = ωL.
This frequency, as you might have already guessed, corresponds to the energy difference between the proton’s two spin states. With the strong magnetic fields generated by the superconducting magnets used in modern NMR instruments, the resonance frequency for protons falls within the radio-wave range, anywhere from 100 MHz to 800 MHz depending on the strength of the magnet.
Attachments
